7.1 缩略词

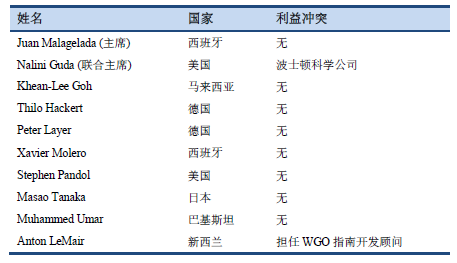
7.2 作者的利益冲突说明
7.3 发表的指南
7.3.1 国际指南
2018 Gut. European evidence-based guidelines on pancreatic cystic neoplasms [29]. Available from: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5890653/
2018 Am J Gastroenterol. ACG clinical guideline: diagnosis and management of pancreatic cysts [49].
2017 J Am Coll Radiol. Management of incidental pancreatic cysts: a white paper of the ACR Incidental Findings Committee [50]. Available from: https://www.jacr.org/article/S1546-1440(17)30318-6/fulltext
2017 Pancreatology. Revisions of international consensus Fukuoka guidelines for the management of IPMN of the pancreas [40].
2015 Gastroenterology. AGA guidelines for the management of pancreatic cysts [43].
2015 World J Gastroenterol. International guidelines for the management of pancreatic intraductal papillary mucinous neoplasms [51]. Available from: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4566378/
2015 Ann Transl Med [Internet]. International consensus on the management of intraductal papillary mucinous neoplasm of the pancreas [45]. Available from: http://www.ncbi.nlm.nih.gov/pmc/articles/PMC4671873/
2013 Dig Liver Dis. European experts consensus statement on cystic tumours of the pancreas [52].
2012 Pancreatology. International consensus guidelines 2012 for the management of IPMN and MCN of the pancreas [8].
2006 Pancreatology. International consensus guidelines for management of intraductal papillary mucinous neoplasms and mucinous cystic neoplasms of the pancreas [53].
7.3.2 地区和其它指南
2015 J Hepatobiliary Pancreat Sci. Revised Japanese guidelines for the management of acute pancreatitis 2015: revised concepts and updated points [54].
2015 Gastroenterology. American Gastroenterological Association technical review on the diagnosis and management of asymptomatic neoplastic pancreatic cysts [55].
2015 Gastroenterology. AGA Institute guideline on the diagnosis and management of asymptomatic neoplastic pancreatic cysts [41].
2015 Gastroenterology. AGA guidelines for the management of pancreatic cysts [43].
2015 Ann Oncol. Cancer of the pancreas: ESMO clinical practice guidelines for diagnosis, treatment and follow-up [56].
2015 Am J Gastroenterol. ACG clinical guideline: genetic testing and management of hereditary gastrointestinal cancer syndromes [57].
2014 Saudi Med J. Saudi Oncology Society clinical management guideline series. Pancreatic cancer [58].
2014 RöFo. S3 guideline for chronic pancreatitis—diagnosis, classification and therapy for the radiologist [59].
2014 Pancreas. American Pancreatic Association practice guidelines in chronic pancreatitis: evidence-based report on diagnostic guidelines [60].
2014 Jpn J Clin Oncol. EBM-based clinical guidelines for pancreatic cancer (2013) issued by the Japan Pancreas Society: a synopsis [61].
2014 Dig Liver Dis. Italian consensus guidelines for the diagnostic work-up and follow-up of cystic pancreatic neoplasms [62].
2014 Diagn Cytopathol. Utilization of ancillary studies in the cytologic diagnosis of biliary and pancreatic lesions: the Papanicolaou Society of Cytopathology guidelines for pancreatobiliary cytology [63].
2014 Diagn Cytopathol. Postbrushing and fine-needle aspiration biopsy follow-up and treatment options for patients with pancreatobiliary lesions: the Papanicolaou Society of Cytopathology guidelines [64].
2014 Diagn Cytopathol. Standardized terminology and nomenclature for pancreatobiliary cytology: the Papanicolaou Society of Cytopathology guidelines [65].
2014 Cancer Cytopathol. Guidelines for pancreaticobiliary cytology from the Papanicolaou Society of Cytopathology: a review [66].
2013 Am J Gastroenterol. American College of Gastroenterology guideline: management of acute pancreatitis [67].
2012 J Natl Compr Canc Netw. Pancreatic adenocarcinoma, version 2.2012: featured updates to the NCCN Guidelines [68].
2012 Ann Oncol. Pancreatic adenocarcinoma: ESMO-ESDO clinical practice guidelines for diagnosis, treatment and follow-up [69].
2008 Ann Oncol. Neuroendocrine gastro-entero-pancreatic tumors: ESMO clinical practice guidelines for diagnosis, treatment and follow-up [70].
2005 Ann Surg Oncol. Treatment guidelines for branch duct type intraductal papillary mucinous neoplasms of the pancreas: when can we operate or observe? [71].
2004 Gastrointest Endosc. ASGE guideline: The role of endoscopy in the diagnosis and the management of cystic lesions and inflammatory fluid collections of the pancreas [72].
Am J Surg Pathol. An illustrated consensus on the classification of pancreatic intraepithelial neoplasia and intraductal papillary mucinous neoplasms [73].
7.4 参考文献
1. Ohno E, Hirooka Y, Kawashima H, Ishikawa T, Kanamori A, Ishikawa H, et al. Natural history of pancreatic cystic lesions: A multicenter prospective observational study for evaluating the risk of pancreatic cancer. J Gastroenterol Hepatol. 2018 Jan;33(1):320–8.
2. Chang YR, Park JK, Jang J-Y, Kwon W, Yoon JH, Kim S-W. Incidental pancreatic cystic neoplasms in an asymptomatic healthy population of 21,745 individuals: Large-scale, single-center cohort study. Medicine (Baltimore). 2016 Dec;95(51):e5535.
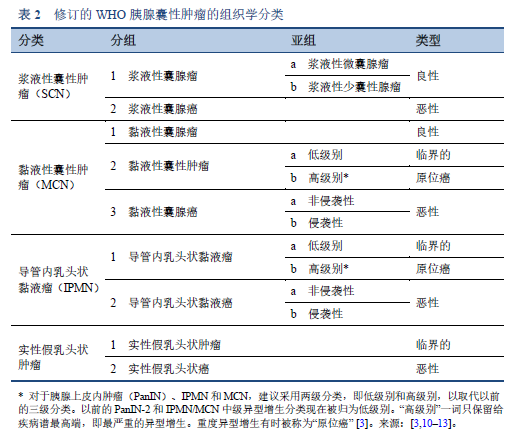
3. Basturk O, Hong S-M, Wood LD, Adsay NV, Albores-Saavedra J, Biankin AV, et al. A revised classification system and recommendations from the Baltimore consensus meeting for neoplastic precursor lesions in the pancreas. Am J Surg Pathol. 2015 Dec;39(12):1730–41.
4. de Pretis N, Mukewar S, Aryal-Khanal A, Bi Y, Takahashi N, Chari S. Pancreatic cysts: diagnostic accuracy and risk of inappropriate resections. Pancreatol Off J Int Assoc Pancreatol IAP Al. 2017 Apr;17(2):267–72.
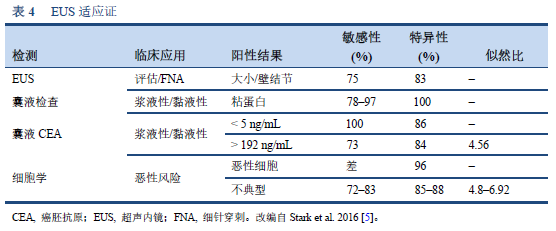
5. Stark A, Donahue TR, Reber HA, Hines OJ. Pancreatic cyst disease: a review. JAMA. 2016 May 3;315(17):1882–93.
6. Gaujoux S, Brennan MF, Gonen M, D’Angelica MI, DeMatteo R, Fong Y, et al. Cystic lesions of the pancreas: changes in the presentation and management of 1,424 patients at a single institution over a 15-year time period. J Am Coll Surg. 2011 Apr;212(4):590–600; discussion 600-603.
7. Karoumpalis I, Christodoulou DK. Cystic lesions of the pancreas. Ann Gastroenterol. 2016 Jun;29(2):155–61.
8. Tanaka M, Fernández-del Castillo C, Adsay V, Chari S, Falconi M, Jang J-Y, et al. International consensus guidelines 2012 for the management of IPMN and MCN of the pancreas. Pancreatology. 2012 Jun;12(3):183–97.
9. Khalid A, Brugge W. ACG practice guidelines for the diagnosis and management of neoplastic pancreatic cysts. Am J Gastroenterol. 2007 Oct;102(10):2339–49.
10. Aaltonen L, Hamilton S, Lambert R. World Health Organization classification of tumours: pathology and genetics of tumours of the digestive system. In: World Health Organization Classification of Tumours. 2000. p. 217–51.
11. Levy MJ. Pancreatic cysts. Gastrointest Endosc. 2009 Feb;69(2 Suppl):S110-116.
12. Lloyd R, Osamura R, Klöppel G, Rosai J. WHO classification of tumours of endocrine organs [Internet]. 4th ed. WHO IARC; 2017 [cited 2018 Apr 27]. 355 p. (IARC WHO Classification of Tumours (Book 10); vol. 10). Available from: http://publications.iarc.fr/Book-And-Report-Series/Who-Iarc-Classification-Of-Tumours/Who-Classification-Of-Tumours-Of-Endocrine-Organs-2017
13. Guilmette JM, Nosé V. Neoplasms of the neuroendocrine pancreas: an update in the classification, definition, and molecular genetic advances. Adv Anat Pathol. 2018 Jun 14;
14. Maire F, Couvelard A, Palazzo L, Aubert A, Vullierme M-P, Rebours V, et al. Pancreatic intraepithelial neoplasia in patients with intraductal papillary mucinous neoplasms: the interest of endoscopic ultrasonography. Pancreas. 2013 Nov;42(8):1262–6.
15. Tsutsumi K, Ohtsuka T, Oda Y, Sadakari Y, Mori Y, Aishima S, et al. A history of acute pancreatitis in intraductal papillary mucinous neoplasms of the pancreas is a potential predictive factor for malignant papillary subtype. Pancreatology. 2010;10(6):707–12.
16. Traverso LW, Moriya T, Hashimoto Y. Intraductal papillary mucinous neoplasms of the pancreas: making a disposition using the natural history. Curr Gastroenterol Rep. 2012 Apr;14(2):106–11.
17. Nilsson LN, Keane MG, Shamali A, Millastre Bocos J, Marijinissen van Zanten M, Antila A, et al. Nature and management of pancreatic mucinous cystic neoplasm (MCN): A systematic review of the literature. Pancreatology. 2016 Dec;16(6):1028–36.
18. Schmid RM, Siveke JT. Approach to cystic lesions of the pancreas. Wien Med Wochenschr 1946. 2014 Feb;164(3–4):44–50.
19. Lévy P, Rebours V. Differential diagnosis of cystic pancreatic lesions including the usefulness of biomarkers. Viszeralmedizin. 2015 Feb;31(1):7–13.
20. Al-Haddad MA, Kowalski T, Siddiqui A, Mertz HR, Mallat D, Haddad N, et al. Integrated molecular pathology accurately determines the malignant potential of pancreatic cysts. Endoscopy. 2015 Feb;47(2):136–42.
21. Morris-Stiff G, Lentz G, Chalikonda S, Johnson M, Biscotti C, Stevens T, et al. Pancreatic cyst aspiration analysis for cystic neoplasms: mucin or carcinoembryonic antigen—which is better? Surgery. 2010 Oct;148(4):638–44; discussion 644-645.
22. Rockacy M, Khalid A. Update on pancreatic cyst fluid analysis. Ann Gastroenterol. 2013;26(2):122–7.
23. Leung KK, Ross WA, Evans D, Fleming J, Lin E, Tamm EP, et al. Pancreatic cystic neoplasm: the role of cyst morphology, cyst fluid analysis, and expectant management. Ann Surg Oncol. 2009 Oct;16(10):2818–24.
24. Brugge WR, Lewandrowski K, Lee-Lewandrowski E, Centeno BA, Szydlo T, Regan S, et al. Diagnosis of pancreatic cystic neoplasms: a report of the cooperative pancreatic cyst study. Gastroenterology. 2004 May;126(5):1330–6.
25. Bick BL, Enders FT, Levy MJ, Zhang L, Henry MR, Abu Dayyeh BK, et al. The string sign for diagnosis of mucinous pancreatic cysts. Endoscopy. 2015 Jul;47(7):626–31.
26. Maker AV, Lee LS, Raut CP, Clancy TE, Swanson RS. Cytology from pancreatic cysts has marginal utility in surgical decision-making. Ann Surg Oncol. 2008 Nov;15(11):3187–92.
27. Hong S-KS, Loren DE, Rogart JN, Siddiqui AA, Sendecki JA, Bibbo M, et al. Targeted cyst wall puncture and aspiration during EUS-FNA increases the diagnostic yield of premalignant and malignant pancreatic cysts. Gastrointest Endosc. 2012 Apr;75(4):775–82.
28. Chiang AL, Lee LS. Clinical approach to incidental pancreatic cysts. World J Gastroenterol. 2016 Jan 21;22(3):1236–45.
29. European Study Group on Cystic Tumours of the Pancreas. European evidence-based guidelines on pancreatic cystic neoplasms. Gut. 2018 May;67(5):789–804.
30. Hackert T, Fritz S, Klauss M, Bergmann F, Hinz U, Strobel O, et al. Main-duct intraductal papillary mucinous nNeoplasm: high cancer risk in duct diameter of 5 to 9 mm. Ann Surg. 2015 Nov;262(5):875–80; discussion 880-881.
31. Morales-Oyarvide V, Mino-Kenudson M, Ferrone CR, Gonzalez-Gonzalez LA, Warshaw AL, Lillemoe KD, et al. Acute pancreatitis in intraductal papillary mucinous neoplasms: A common predictor of malignant intestinal subtype. Surgery. 2015 Nov;158(5):1219–25.
32. Konings ICAW, Harinck F, Poley J-W, Aalfs CM, van Rens A, Krak NC, et al. Prevalence and progression of pancreatic cystic precursor lesions differ between groups at high risk of developing pancreatic cancer. Pancreas. 2017 Jan;46(1):28–34.
33. Capurso G, Boccia S, Salvia R, Del Chiaro M, Frulloni L, Arcidiacono PG, et al. Risk factors for intraductal papillary mucinous neoplasm (IPMN) of the pancreas: a multicentre case-control study. Am J Gastroenterol. 2013 Jun;108(6):1003–9.
34. Ohtsuka T, Kono H, Nagayoshi Y, Mori Y, Tsutsumi K, Sadakari Y, et al. An increase in the number of predictive factors augments the likelihood of malignancy in branch duct intraductal papillary mucinous neoplasm of the pancreas. Surgery. 2012 Jan;151(1):76–83.
35. Perez-Johnston R, Narin O, Mino-Kenudson M, Ingkakul T, Warshaw AL, Fernandez-Del Castillo C, et al. Frequency and significance of calcification in IPMN. Pancreatology. 2013 Feb;13(1):43–7.
36. Yagi Y, Masuda A, Zen Y, Takenaka M, Toyama H, Sofue K, et al. Predictive value of low serum pancreatic enzymes in invasive intraductal papillary mucinous neoplasms. Pancreatol Off J Int Assoc Pancreatol IAP Al. 2016 Oct;16(5):893–9.
37. Fritz S, Hackert T, Hinz U, Hartwig W, Büchler MW, Werner J. Role of serum carbohydrate antigen 19-9 and carcinoembryonic antigen in distinguishing between benign and invasive intraductal papillary mucinous neoplasm of the pancreas. Br J Surg. 2011 Jan;98(1):104–10.
38. Chang Y-T, Tien Y-W, Jeng Y-M, Yang C-Y, Liang P-C, Wong J-M, et al. Overweight increases the risk of malignancy in patients with pancreatic mucinous cystic neoplasms. Medicine (Baltimore). 2015 May;94(20):e797.
39. Lafemina J, Katabi N, Klimstra D, Correa-Gallego C, Gaujoux S, Kingham TP, et al. Malignant progression in IPMN: a cohort analysis of patients initially selected for resection or observation. Ann Surg Oncol. 2013 Feb;20(2):440–7.
40. Tanaka M, Fernández-del Castillo C, Kamisawa T, Jang JY, Levy P, Ohtsuka T, et al. Revisions of international consensus Fukuoka guidelines for the management of IPMN of the pancreas. Pancreatology. 2017 Sep;17(5):738–53.
41. Vege SS, Ziring B, Jain R, Moayyedi P, Clinical Guidelines Committee, American Gastroenterology Association. American Gastroenterological Association institute guideline on the diagnosis and management of asymptomatic neoplastic pancreatic cysts. Gastroenterology. 2015 Apr;148(4):819–22; quize12-13.
42. Sahani DV, Kambadakone A, Macari M, Takahashi N, Chari S, Fernandez-del Castillo C. Diagnosis and management of cystic pancreatic lesions. AJR Am J Roentgenol. 2013 Feb;200(2):343–54.
43. Lennon AM, Ahuja N, Wolfgang CL. AGA guidelines for the management of pancreatic cysts. Gastroenterology. 2015 Sep;149(3):825.
44. Heckler M, Michalski CW, Schaefle S, Kaiser J, Büchler MW, Hackert T. The Sendai and Fukuoka consensus criteria for the management of branch duct IPMN—a meta-analysis on their accuracy. Pancreatology. 2017 Mar;17(2):255–62.
45. Tanaka M. International consensus on the management of intraductal papillary mucinous neoplasm of the pancreas. Ann Transl Med. 2015 Nov;3(19):286.
46. Choi SH, Park SH, Kim KW, Lee JY, Lee SS. Progression of unresected intraductal papillary mucinous neoplasms of the pancreas to cancer: a systematic review and meta-analysis. Clin Gastroenterol Hepatol. 2017 Oct;15(10):1509-1520.e4.
47. Crippa S, Pezzilli R, Bissolati M, Capurso G, Romano L, Brunori MP, et al. Active surveillance beyond 5 years is required for presumed branch-duct intraductal papillary mucinous neoplasms undergoing non-operative management. Am J Gastroenterol. 2017 Jul;112(7):1153–61.
48. Anand N, Sampath K, Wu BU. Cyst features and risk of malignancy in intraductal papillary mucinous neoplasms of the pancreas: a meta-analysis. Clin Gastroenterol Hepatol. 2013 Aug;11(8):913–21; quiz e59-60.
49. Elta GH, Enestvedt BK, Sauer BG, Lennon AM. ACG clinical guideline: diagnosis and management of pancreatic cysts. Am J Gastroenterol. 2018;113(4):464–79.
50. Megibow AJ, Baker ME, Morgan DE, Kamel IR, Sahani DV, Newman E, et al. Management of incidental pancreatic cysts: a white paper of the ACR Incidental Findings Committee. J Am Coll Radiol JACR. 2017 Jul;14(7):911–23.
51. Goh BK. International guidelines for the management of pancreatic intraductal papillary mucinous neoplasms. World J Gastroenterol WJG. 2015 Sep 14;21(34):9833–7.
52. Del Chiaro M, Verbeke C, Salvia R, Klöppel G, Werner J, McKay C, et al. European experts consensus statement on cystic tumours of the pancreas. Dig Liver Dis. 2013 Sep;45(9):703–11.
53. Tanaka M, Chari S, Adsay V, Fernandez-del Castillo C, Falconi M, Shimizu M, et al. International consensus guidelines for management of intraductal papillary mucinous neoplasms and mucinous cystic neoplasms of the pancreas. Pancreatology. 2006;6(1–2):17–32.
54. Isaji S, Takada T, Mayumi T, Yoshida M, Wada K, Yokoe M, et al. Revised Japanese guidelines for the management of acute pancreatitis 2015: revised concepts and updated points. J Hepato-Biliary-Pancreat Sci. 2015 Jun;22(6):433–45.
55. Scheiman JM, Hwang JH, Moayyedi P. American Gastroenterological Association technical review on the diagnosis and management of asymptomatic neoplastic pancreatic cysts. Gastroenterology. 2015 Apr;148(4):824-848.e22.
56. Ducreux M, Cuhna AS, Caramella C, Hollebecque A, Burtin P, Goéré D, et al. Cancer of the pancreas: ESMO Clinical Practice Guidelines for diagnosis, treatment and follow-up. Ann Oncol Off J Eur Soc Med Oncol. 2015 Sep;26 Suppl 5:v56-68.
57. Syngal S, Brand RE, Church JM, Giardiello FM, Hampel HL, Burt RW, et al. ACG clinical guideline: genetic testing and management of hereditary gastrointestinal cancer syndromes. Am J Gastroenterol. 2015 Feb;110(2):223–62; quiz 263.
58. Rahal MM, Bazarbashi SN, Kandil MS, Al-Shehri AS, Alzahrani AM, Aljubran AH, et al. Saudi Oncology Society clinical management guideline series. Pancreatic cancer 2014. Saudi Med J. 2014 Dec;35(12):1534–7.
59. Schreyer AG, Jung M, Riemann JF, Niessen C, Pregler B, Grenacher L, et al. S3 guideline for chronic pancreatitis—diagnosis, classification and therapy for the radiologist. RöFo. 2014 Nov;186(11):1002–8.
60. Conwell DL, Lee LS, Yadav D, Longnecker DS, Miller FH, Mortele KJ, et al. American Pancreatic Association practice guidelines in chronic pancreatitis: evidence-based report on diagnostic guidelines. Pancreas. 2014 Nov;43(8):1143–62.
61. Yamaguchi K, Okusaka T, Shimizu K, Furuse J, Ito Y, Hanada K, et al. EBM-based clinical guidelines for pancreatic cancer (2013) issued by the Japan Pancreas Society: a synopsis. Jpn J Clin Oncol. 2014 Oct;44(10):883–8.
62. Italian Association of Hospital Gastroenterologists and Endoscopists, Italian Association for the Study of the Pancreas, Buscarini E, Pezzilli R, Cannizzaro R, De Angelis C, et al. Italian consensus guidelines for the diagnostic work-up and follow-up of cystic pancreatic neoplasms. Dig Liver Dis. 2014 Jun;46(6):479–93.
63. Layfield LJ, Ehya H, Filie AC, Hruban RH, Jhala N, Joseph L, et al. Utilization of ancillary studies in the cytologic diagnosis of biliary and pancreatic lesions: the Papanicolaou Society of Cytopathology guidelines for pancreatobiliary cytology. Diagn Cytopathol. 2014 Apr;42(4):351–62.
64. Kurtycz D, Tabatabai ZL, Michaels C, Young N, Schmidt CM, Farrell J, et al. Postbrushing and fine-needle aspiration biopsy follow-up and treatment options for patients with pancreatobiliary lesions: the Papanicolaou Society of Cytopathology guidelines. Diagn Cytopathol. 2014 Apr;42(4):363–71.
65. Pitman MB, Centeno BA, Ali SZ, Genevay M, Stelow E, Mino-Kenudson M, et al. Standardized terminology and nomenclature for pancreatobiliary cytology: the Papanicolaou Society of Cytopathology guidelines. Diagn Cytopathol. 2014 Apr;42(4):338–50.
66. Pitman MB, Layfield LJ. Guidelines for pancreaticobiliary cytology from the Papanicolaou Society of Cytopathology: A review. Cancer Cytopathol. 2014 Jun;122(6):399–411.
67. Tenner S, Baillie J, DeWitt J, Vege SS, American College of Gastroenterology. American College of Gastroenterology guideline: management of acute pancreatitis. Am J Gastroenterol. 2013 Sep;108(9):1400–15; 1416.
68. Tempero MA, Arnoletti JP, Behrman SW, Ben-Josef E, Benson AB, Casper ES, et al. Pancreatic adenocarcinoma, version 2.2012: featured updates to the NCCN Guidelines. J Natl Compr Cancer Netw JNCCN. 2012 Jun 1;10(6):703–13.
69. Seufferlein T, Bachet JB, Van Cutsem E, Rougier P, ESMO Guidelines Working Group. Pancreatic adenocarcinoma: ESMO-ESDO clinical practice guidelines for diagnosis, treatment and follow-up. Ann Oncol. 2012 Oct;23 Suppl 7:vii33-40.
70. Öberg K, Knigge U, Kwekkeboom D, Perren A, ESMO Guidelines Working Group. Neuroendocrine gastro-entero-pancreatic tumors: ESMO Clinical Practice Guidelines for diagnosis, treatment and follow-up. Ann Oncol. 2012 Oct;23 Suppl 7:vii124-130.
71. Jang J-Y, Kim S-W, Lee SE, Yang SH, Lee KU, Lee YJ, et al. Treatment guidelines for branch duct type intraductal papillary mucinous neoplasms of the pancreas: when can we operate or observe? Ann Surg Oncol. 2008 Jan;15(1):199–205.
72. Jacobson BC, Baron TH, Adler DG, Davila RE, Egan J, Hirota WK, et al. ASGE guideline: the role of endoscopy in the diagnosis and the management of cystic lesions and inflammatory fluid collections of the pancreas. Gastrointest Endosc. 2005 Mar;61(3):363–70.
73. Hruban RH, Takaori K, Klimstra DS, Adsay NV, Albores-Saavedra J, Biankin AV, et al. An illustrated consensus on the classification of pancreatic intraepithelial neoplasia and intraductal papillary mucinous neoplasms. Am J Surg Pathol. 2004 Aug;28(8):977–87.